In June 1967, at Maimonides Hospital in Brooklyn, New York, Dr. Adrian Kantrowitz and his surgical team successfully used a new device—the intra-aortic balloon pump (IABP)—to treat a patient in cardiogenic shock. This small device was an exciting innovation. It assisted the failing left ventricle while the natural heart remained in place, and it was nonobligatory; i.e., it could be turned on and off repeatedly without adverse effects. The IABP had been designed and fabricated within 12 months by Kantrowitz and his surgical-engineering research team at Maimonides. The research was funded by a National Heart Institute (NHI) program-project grant (PPG) designed to support the work from concept and design to animal studies to clinical use, in effect a translational medicine program-project. The rapid development of the IABP was facilitated by Kantrowitz’s earlier NHI-funded investigations, which included a left ventricular assist device (LVAD) intended as “destination therapy” for chronic heart failure.
Sixteen patients were treated with the IABP during 1967–1968. After Kantrowitz published his initial results, teams of cardiologists and cardiac surgeons from more than 40 countries visited the Assisted Circulation Research Laboratory at Maimonides. All were eager to learn more about this promising new medical technology. The dramatic success of the phase-shift balloon pump, as it was then called, prompted Kantrowitz to organize a cooperative study, including nine leading cardiovascular centers, to validate the initial clinical experience with an intra-aortic, counterpulsating LVAD.
Yet, in May 1970, just 3 years following the balloon pump’s use in the first patient, the New York Times reported that Dr. Adrian Kantrowitz, his entire surgical resident and research staff and their families, 25 in all, would be relocating to Detroit, Michigan. Why the exodus? The Editor-in-Chief of this journal invited this writer (who had tended to the administrative work of the move) to share her recollection. He told her, “Everyone knew of the en masse departure, but not why it happened.” It is likely that no one ever had all of the facts, but archived documents allow some informed conjecture about this event. Oddly enough, the answer seems to center on a National Institutes of Health (NIH) grant mechanism, NIH policy with respect to institutional sponsor obligations, as well as competing collegial ambitions in a community hospital.
A Look Back …
Adrian Kantrowitz joined the Maimonides Hospital* medical staff as full-time director of cardiovascular surgery in 1955, just 3 years after Dr. John Gibbon used the pump oxygenator to perform the first successful open heart surgery 1. In 1952, Kantrowitz had been a US Public Health Service Research Fellow in the cardiovascular physiology laboratory of Dr. Carl Wiggers at Western Reserve University in Cleveland. In the Wiggers Laboratory, Adrian Kantrowitz conducted experiments to study the feasibility of his idea about a method to increase coronary flow by perfusing “… the coronary bed with systolic pressures during myocardial diastole, so as to be able to take advantage of the markedly decreased peripheral resistance during this period.” He envisioned accomplishing this “… by delaying the arterial pressure peak long enough so that it would occur during diastole” (Fig. 1) 2.
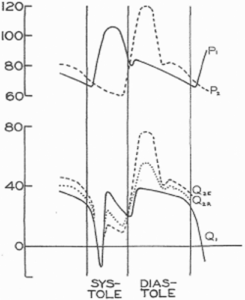
Figure 1
Heavy lines indicate normal aortic pressure (P1) and phasic coronary flow (Q1). Dashed and dotted lines indicate predicted flows when the anterior descending coronary artery is perfused with pulse pressure out of phase with myocardial systole. P2 represents proposed delayed coronary pressure; Q2R, calculated flow in presumed rigid coronary system; Q2E, calculated flow in presumed elastic coronary system 2.
Adrian’s brother, Arthur, a professor of physics at the Cornell University Graduate School of Aeronautical Engineering, lent his expertise in fluid dynamics, brought analytic support for the experimental data, and acted as a critical sounding board for the project. Most important was the environment: in the Wiggers Laboratory, where Kantrowitz was surrounded by some of the best investigators in cardiovascular physiology. To implement the concept of diastolic augmentation—later popularized by Dwight Harken as “counterpulsation” 3, 4—Kantrowitz first explored a biological approach. Intrigued by the idea of powering the system with “the eggs eaten for breakfast” 5, he wrapped a leaf of the diaphragm around the descending aorta and stimulated the phrenic nerve during diastole 6.
Upon his full-time appointment at Maimonides, Kantrowitz had applied for and was awarded three successive NIH grants ranging from $9000 to $15 000 a year for “Augmentation of Coronary Flow” and “Experimental Construction of an Auxiliary Ventricle.” During 1958–1960, Kantrowitz visited the NHI to discuss prospects for future funding. Dr. Robert Ringler, Deputy Director of the NHI, met with Kantrowitz, listened to his vision of “leaving the natural heart in place and fashioning an assist pump from autologous muscle.” Ringler was convinced. He said, “Adrian, you need a program-project grant (PPG), but that’s too big a leap from a $15 000 grant. Apply first for a multiproject grant for $200 000 to $300 000. If you manage that successfully, you’ll be taken seriously for leadership of a program-project.”
Following Dr. Ringler’s advice, in 1962, Kantrowitz obtained a 3-year, multiproject grant, “Integrated Electronic Control of Physiologic Systems,” proposing therapeutic applications using prolonged neurostimulation. In acute animal experiments, he was able to demonstrate the feasibility of stimulating the phrenic nerve to contract the aorta during diastole 6. The neuromuscular stimulation experiments led to a cluster of feasibility studies for treatment of conditions such as neurogenic bladder and paralytic ileus 7, 8. Among these was the first demonstration of a paraplegic standing and sitting under computer control 9. However, the prerequisite long-term stimulation of the phrenic nerve resulted in scar formation, a formidable challenge. Kantrowitz shifted to a mechanical device 10, 11. Work on muscle-powered cardiac assistance was picked up later as cardiomyoplasty by Stevenson, Macoviak, and others 12.
In 1965, Ringler invited Kantrowitz to submit a PPG application. The PPG would be called “Studies in Assisted Circulation.” Later that same year, at its August meeting, the Maimonides Research Committee discussed the need for a General Clinical Research Center (CRC) funded by NIH. Kantrowitz’s proposed program project was for a Categorical CRC studying mechanical methods of assisting the failing heart. It included a patient study unit. The Maimonides Research Committee Chair, distinguished hematologist Dr. Stanley Lee, was convinced (or had been so advised) that the NIH was not likely to fund two clinical research centers in a community hospital such as Maimonides. Hence, if funded, the Kantrowitz Categorical PPG, with its dedicated cardiac assist patient study unit, would foreclose an NIH-funded General CRC unit at Maimonides.
During the August 1965 Research Committee Meeting, Kantrowitz argued strongly that his program project grant would be easier to obtain than one for a General Clinical Research Center, partly because it had been invited by the NHI and partly because of the well-defined focus at the NHI on new methods of treating heart failure. He told the Research Committee that he and his staff were making an intense effort on a proposal to organize a vertical, assisted circulation research program: from concept, bench, and animal testing, through human trial. Kantrowitz had already mobilized the Chairs of each relevant specialty, including hematology at Maimonides. Each Chair participated in writing the application. Each led subprojects to answer questions on the physiological and psychological impact of prolonged mechanical circulatory assistance. Nephrologists would study renal function; hematologists, hemostatic mechanisms; psychiatrists, the reaction of recipients to living with a life-sustaining mechanical implant; and so on.
Importantly, the application also requested funds for a dedicated two-bed cardiac assist device patient study unit. It would be equipped with an advanced, custom-designed monitoring and recording system, initially, for intensive study of one patient at a time. It was, in fact, a translational medicine program project application.
Kantrowitz asked the Maimonides Research Committee to consider submitting the General and Categorical Clinical Research Center applications sequentially. A majority of the Committee concurred. The Research Committee approved Kantrowitz’s PPG application for submission to NIH by Maimonides Hospital in September 1966.
Soon after the PPG application was submitted to NIH, Research Committee Chair, Dr. Lee, sent Kantrowitz a draft CRC grant application prepared for submission to the NIH. This draft CRC application included, verbatim, the mechanical circulatory assist research plan proposed for Maimonides in Kantrowitz’s PPG application, already pending at NIH. Kantrowitz replied to Dr. Lee saying that the inclusion would be acceptable (as would be expected at NIH) “if the assisted circulation section was clearly identified as part of the pending categorical PPG application ‘Studies in Assisted Circulation’.” Dr. Lee refused, and then asked Kantrowitz to withdraw the PPG application in favor of the CRC application. Kantrowitz refused.
As far as is known, the CRC application was not submitted. The PPG was funded for 5 years, 1967–1972, in the amount of $3 million. The envisioned Categorical 2-bed Study Research Center was built during 1968, and opened in January 1969 (Fig. 2). Dr. Theodore Cooper, Director of the NHI, welcomed guests at the opening.

Figure 2
The Heart Surgery Study Unit at Maimonides Medical Center, funded by an NIH program-project grant, opened in January 1969. Dr. Dov Jaron, bioengineer and Director of the Assisted Circulation Research Laboratory, is seated at the custom-designed Gulton Instruments monitoring and recording system. The Unit’s Research Nurse, Cleta Lane, is at the bedside.
A “Destination” LVAD Cross-Fertilized to a Temporary LVAD: The IABP
With Kantrowitz’s earlier multiproject grant, the Assisted Circulation Research team at Maimonides had designed and fabricated an avalvular, U-shaped auxiliary ventricle implanted by means of short grafts attached to the divided ascending aorta (Figs. 3 and 4) 13. Prolonged experiments in animals with induced heart failure yielded promising results 14. To prepare for human use, which involved scaling up the canine sized blood pump and designing and building a more robust drive unit, Kantrowitz again enlisted his brother, Arthur. Arthur Kantrowitz, by then director of the AVCO-Everett Aerospace Research Laboratory in Massachusetts, brought the AVCO-Everett team of outstanding engineers into the effort. This fraternal collaboration culminated in an interruptible, left ventricular assist device (LVAD or VAD) (Figs. 5–7). It was counterpulsating, based on the concept demonstrated more than a decade earlier in the Wiggers Laboratory.
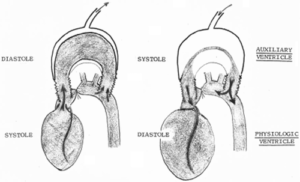
Figure 3
Drawing of counterpulsation concept for devices used in preclinical experiments at the Maimonides Surgical Research Laboratory. During diastole of auxiliary ventricle (AV), the bulb fills with blood as air is removed by suction, lowering resistance of the left ventricle to emptying and thus reducing its work. During AV systole, air contracts the bulb that expels its blood into the arterial tree, increasing diastolic pressure as well as peripheral and coronary flow 13.
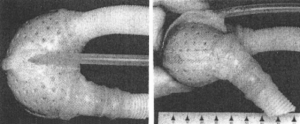
Figure 4
Two views of an early prototype of the silastic auxiliary ventricle showing the angle of inset arms and relative length of woven Dacron cuffs 13.

Figure 6
Adrian Kantrowitz and his brother, physicist, Arthur Kantrowitz collaborated on the U-shaped auxiliary ventricle.
Courtesy of Ralph Morse / LIFE Picture Collection / Getty Images.
Figure 7
Dr. Tetsuzu Akutsu, Director of Experimental Surgery, and Dr. Adrian Kantrowitz discussing a preclinical experiment with the U-shaped auxiliary ventricle.
Courtesy of Ralph Morse / LIFE Picture Collection / Getty Images.
In May 1966 at Maimonides, a scaled-up U-shaped mechanical auxiliary ventricle made by the AVCO-Everett Laboratory was implanted in a 63-year-old woman, Case L(VAD)-1, in terminal heart failure. The auxiliary ventricle was intended for long-term, ambulatory treatment (“destination therapy”) of chronic heart failure. The patient could be ambulatory, detached from a drive unit, because of the blood pump’s nonobligatory function. It could be turned on and off, and on again, safely.
Case L-1 responded well to the pump hemodynamically. She was taken out of heart failure when the pump was activated. Her heart failure returned when the pump was deactivated. However, during surgery, it had been necessary to lengthen the blood pump grafts for attachment to the aorta. On postoperative day 12, Case L-1 suffered a stroke due to thromboemboli emanating from the lengthened grafts, and died the following day 15. She was the first patient to be implanted with an assist device intended to remain in the body after discharge from the hospital, when the patient returned home.
While the first Kantrowitz LVAD was being developed, the NIH, in 1964, created an Advisory Group to focus on initiatives for treatment of the failing heart. The Advisory Council of NIH recommended that NIH prioritize the development of an artificial heart. The NHI commissioned six studies on the Feasibility of a Total Artificial Heart. In 1966, Arthur Kantrowitz’s AVCO-Everett group prepared one of these reports. Under a subcontract, Adrian’s Maimonides team wrote the biomedical section. The Feasibility Studies called attention to the need for a “family of temporary emergency devices, long-term partial heart replacement devices, and total artificial hearts” for the progressive stages of heart failure 16.
In preparing their report, both Adrian and Arthur Kantrowitz were freshly impressed by the huge unmet need for temporary assistance to the acutely failing left ventricle. Each also recognized how their recent joint work on the U-shaped auxiliary ventricle for chronic left ventricular (LV) failure demonstrated the potential of counterpulsation for treatment of acute LV failure. They were both aware of the work published by Moulopoulos and Clauss (of the Kolff and Harken groups, respectively) on an IABP. These two groups had published on animal and cadaver studies but not studies in human subjects 17, 18.
Because tensions between Adrian and Arthur Kantrowitz had grown during their work together, the brothers decided to pursue the IABP separately. Adrian had the PPG funding and the multidisciplinary team in place in his Maimonides surgical research laboratory. Arthur joined with cardiac surgeons, Dr. Gerald Austen and Dr. Mortimer Buckley, at Massachusetts General Hospital 19. Paul Freed and Dr. Wladimir Schilt, a Surgical Research Associate in the Maimonides Surgical Research Laboratory, cross-fertilized their work on the U-shaped auxiliary ventricle to design and preclinically test the IABP in animals within 12 months 20. For human use, the device required only the Hospital’s Research Committee’s approval. That approval was obtained in early June 1967. Within weeks, the investigational device was used in Case 1.
Case 1
At 5 o’clock the morning of Tuesday, June 29, 1967, a 45-year-old woman (Case 1) was brought to the Emergency Room at Maimonides Medical Center (MMC) of Brooklyn. Dr. Menachem Shapiro, the Internal Medicine Chief Resident, examined her. She had chest pain, was agitated, dyspneic, cyanotic, diaphoretic, pale, cold, and clammy. An ECG suggested an acute myocardial infarction. She did not respond to the maximal available medical treatment of the day: Levophed, ethacrynic acid, Solu-Medrol, and Isuprel. Her blood pressure did not rise above 60/30. Her central venous pressure rose to 14. Her medical chart notes: “cardiovascular collapse, her condition grave.”
Aware that the Hospital’s Medical Research Committee had recently approved Kantrowitz’s protocol for human trial of a new cardiac assist device to treat patients in cardiogenic shock, Dr. Shapiro called Dr. Steven Phillips. Dr. Phillips, then in his last month of surgical internship, quickly recognized that this patient might indeed be an appropriate candidate for the phase shift balloon pump (Fig. 8). At 6 am, she was admitted to the hospital. At 8:50 am, she was examined by Dr. Walter Feder, Chief of Cardiology. She was anuric. Her blood pressure was 70/30 on Levophed. At 9:20 am, she was described as “shocky.” Dr. Kantrowitz and Dr. Jordan Haller, Director of Thoracic Surgery, had joined Dr. Phillips and Dr. Feder at bedside. At 10:30 am, the team agreed that this patient met the protocol criteria. Paul Freed, a young bioengineer who co-designed the phase-shift balloon pump system, operated the drive unit. Dr. Kantrowitz asked Dr. Phillips to insert the balloon pump. Dr. Phillips inserted the balloon pump (Fig. 9) through the patient’s femoral artery, clamping it below the balloon pump insertion site, as was then specified in the protocol. At 11:32 am, cardiac assistance was begun. All vasopressors were stopped and 75 mg i.v. heparin was started. By 11:45 am, Case 1 was improving: urine output resumed; her color was better; she was no longer cold and clammy. By 12:15 pm, she was alert and drank 6 ounces of water. She was no longer in shock. At 1:00 pm, the balloon pump was turned off; labs were drawn. The patient became “very agitated,” and balloon pumping was resumed. By 1:15 pm, her condition was improved. At 2 pm, the balloon pump was turned off. Again, she became agitated. By 2:30 pm, her blood pressure was falling, and the balloon pump was restarted. The patient’s systolic and diastolic pressures gradually increased. Her urinary output increased from zero to an average of 40 cc/h. Her skin became warm and dry. The balloon pump was turned off at 6:24 pm. At 8:00 am, the balloon pump was withdrawn. It had been activated intermittently nine times, throughout 7 hours 21.
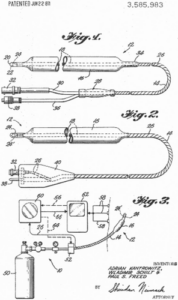
Figure 8
U.S. Patent 3 585 983. Inventors: Adrian Kantrowitz; Wladimir Schilt; Paul S. Freed, all of c/o Maimonides Medical Center 4802 Tenth St., Brooklyn, N.Y. 11219. Filed Mar. 5, 1968, Patented June 22, 1971.
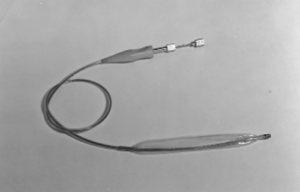
Figure 9
Intraaortic balloon pump made for the Cooperative Clinical trial. The pressure sensor is at the tip of the balloon. Two EKG electrodes, one at each end of the balloon, are incorporated.
This first patient remained in the hospital for observation and was discharged 3 months later on September 17, 1967. In the following months, she was seen as an outpatient. She was in good health, except for a significant limp, related to clamping of the femoral artery during balloon pump activation. In January 1969, she was killed in an auto accident, some 18 months after she recovered from cardiogenic shock with assistance from the IABP.
Dr. Kantrowitz Is Not a Branch Store of the NIH …
By 1968, 1 year after the first IABP patient was successfully treated for cardiogenic shock, the Kantrowitz team had treated 16 patients using the balloon pump 22. The team published encouraging results, exciting global interest in the cardiovascular community (Figs. 10, 11). Cardiologists and cardiac surgeons from more than 40 countries visited the Maimonides laboratory (Figs. 12 and 13). Industry executives from device companies including Baxter, Datascope, Bard, and Ethicon came to observe animal experiments. Many visitors came repeatedly. Eager to use the balloon pump, they asked how to obtain the equipment. The Kantrowitz group had designed and fabricated the balloon pump catheters in the surgical research laboratory at Maimonides. At first, Paul Freed modified a dual-beam Tektronix oscilloscope to use as a drive unit. Later, IABP drive units were designed with the bioengineering group at Rutgers University led by Professors Walter Welkowitz and Donald A. Molony (Fig. 14) 23. No balloon pump systems were commercially available. Kantrowitz was convinced that the balloon pump’s effects needed validation by others. He wrote to Dr. Ringler asking permission to supply IABP systems to a few investigators whose institutions would reimburse funded PPG. Ringler responded stonily, “Adrian, you were peer reviewed. Your investigators were not.” Mincing no words, Dr. Ringler was informing Dr. Kantrowitz that he was not a branch store of the NIH.

Figure 10

Figure 11
Figure 12

Figure 13

Figure 14
A Multicenter Feasibility Trial
Kantrowitz’s next thought was to organize a multicenter study. The Cooperative Study would obtain independent funding to cover contracted manufacture of the equipment and other study expenses. To explore the possibility, Kantrowitz invited nine of the most interested and qualified investigators. He assumed that half might want to participate. All nine wanted in (Fig. 15). To enable face-to-face meetings, Kantrowitz obtained a $23 000 planning grant from the local David and Minnie Berk Foundation. Hedging their bet, the investigators decided to submit two parallel applications for funding: one to the NIH and one to the John A. Hartford Foundation.

Figure 15
At a March 1969 meeting in Bethesda, the Cooperative Study Investigators discussed their proposed project to study the physiologic effects of diastolic augmentation with Dr. Theodore Cooper, Director of the NHI and his colleague, Dr. Sheila Mitchell. Dr. Mitchell had visited Maimonides and had seen the IABP patients first-hand. Emphasizing that the final decision would be made by peer reviewers, both NHI officials argued strongly for a randomized study design. At the Bethesda meeting, the investigators opposed a randomized study design at this stage. Dr. Thomas Killip cited the mortality of cardiogenic shock patients at Cornell’s Myocardial Infarction Research Unit (MIRU) of which he was Principal Investigator: 75–85% percent, and virtually 100%, when the patient had systolic blood pressure of less than 70 mm Hg in the presence of acute myocardial infarction. Similar outcomes prevailed at the three other MIRUs represented whose Principal Investigators had joined the Cooperative Study: Duke University, the University of Chicago, and Cedars-Sinai Hospital.
Much had been written about mortality in cardiogenic shock. However, the lack of hard data was actually the impetus for NHI to establish the MIRU program. Given the dismal outlook of cardiogenic shock with standard medical management, the investigators argued that randomization was unethical and premature. They wanted to first familiarize themselves with the IABP system, its counterintuitive medical management 24, and the physiologic effects of diastolic augmentation in cardiogenic shock patients. “A randomized study could follow,” they noted. The investigators designated a small Steering Committee to control the study, St. Vincent’s Medical Center in New York City as the Data Coordinating Center, and MMC as the sponsor for administering funds and ensuring standardized use of the IABP system among centers.
The Research Committees of each of the nine other participating centers approved the application. As the sponsor, Maimonides was the last to consider the application. The Cooperative Study grant application was approved by the Maimonides Research Committee and submitted to NIH by the hospital on June 11, 1969.
Two Weeks Later
On June 26, 1969, the MMC Research Committee Chair, Dr. Lee, notified Kantrowitz that a two-person Ad Hoc Research Subcommittee would be reviewing all balloon pump study patients treated at Maimonides. Dr. Lee had also appointed himself Chair of the Ad Hoc Research Subcommittee. The second member of the Ad Hoc Subcommittee was the Department Chair of Pediatrics, Dr. Howard Joos, who had gone head-to-head in a bitter argument with Kantrowitz during the 1966 infant heart transplant planning sessions. A highly respected clinician, Dr. Joos wanted to remove the heart of the doomed anencephalic donor infant after death was declared. Kantrowitz wanted to remove the heart while it was still viable 25, 26. Kantrowitz eventually conceded.
The Ad Hoc Subcommittee reviewed the records of 30 balloon pump study patients. It was standard procedure for all Ad Hoc Subcommittees to meet with the Principal Investigator regarding questions or concerns prior to the full Research Committee meeting. In this case, the Subcommittee made no attempt to have such a meeting with Kantrowitz. Given that 2 weeks earlier, the Research Committee had approved the Cooperative Study protocol, which already met many of the Subcommittee’s criticisms, Kantrowitz assumed that the Subcommittee members had no questions. Several days before the August 1 Research Committee meeting, Kantrowitz was given a list of criticisms from the Ad Hoc Subcommittee.
The Triggering Event …
At the August 1, 1969 meeting of the Maimonides Research Committee, Kantrowitz and Joseph Krakauer, the surgical intensivist who had attended every IABP study patient, were present. Michael Meyer, a laboratory staff member, took detailed notes. The Ad Hoc Subcommittee report included numerous criticisms of the balloon pump study: departures from the protocol; absence of cardiologists for the decision to use IABP; adverse events caused by the IABP (including limb ischemia resulting from clamping the femoral artery for balloon pump insertion and myocardial ruptures causing death); and “to have a good cooperative study, there has to be randomization of case selection.”
Kantrowitz addressed the myocardial ruptures. The team had studied all patients including those who had suffered myocardial ruptures. It was found that the patients fell into two groups—those in whom shock developed relatively early, less than 36 hours after myocardial infarction, and those in whom the interval from the infarction to shock was longer. Kantrowitz said that all of the myocardial ruptures occurred in the “longer interval” group, leading to the assessment that the balloon pump had sustained these patients long enough to enable “the natural evolution of massive myocardial” lesions to be completed 27. Regarding the initial balloon pump insertion-procedure of clamping the femoral artery, he noted that it had already been replaced by a revised graft procedure, approved by the MMC Research Committee. This graft procedure had been implemented in all patients, subsequent to the first. The revised procedure had been published 1 year earlier, in 1968 in the Journal of Thoracic and Cardiovascular Surgery 28.
The Ad Hoc Subcommittee also stated that the cardiologists designated to be present for the decision to enter each patient in the study were largely absent. The contact log revealed that in those instances, the designated cardiologist was himself the referring physician. The few exceptions occurred when the patient was deteriorating rapidly.
Kantrowitz pointed out an inherent difficulty for a randomized study of IABP: vasopressors were standard medical management of acute heart failure, whereas vasodilators were the adjunctive management of diastolic augmentation. He reiterated the position taken by the Cooperative Study Investigators during the meeting at NIH: the first stage should be gaining familiarity with the device and its adjunctive management.
Proceeding to a vote, the Research Committee accepted the Ad Hoc Subcommittee’s recommendation to suspend the ongoing trial of IABP at MMC. The Research Committee also voted that the Hospital withdraw the Cooperative Study application pending at NHI. Kantrowitz was informed that he could appeal to the Executive Council of the Hospital but that the Hospital’s Research Committee was considered final authority by the NIH. Maimonides Hospital then formally withdrew the Cooperative Study application pending at NIH. It was clear to Kantrowitz that it was untenable for him to remain at MMC. On January 1, 1970, he submitted his resignation, effective June 30, 1970.
The Fallout
A house staff revolt …
When Kantrowitz’s intended departure became known, the entire surgical resident staff announced they would not remain at MMC. They had come to MMC because of Dr. Kantrowitz; they wanted to go wherever he would go. MMC was a teaching affiliate of State University of New York (SUNY) Downstate. The eight surgical residents, all under contract with MMC, turned to Dr. Clarence Dennis, Chair of Surgery at the SUNY Downstate Medical Center, for advice. Dr. Dennis, a pioneer cardiovascular surgeon himself, was ill during the Research Committee actions. Upon returning to work, he came to Maimonides for a full day to review the chart of every IABP study patient. Dr. Dennis concluded that the Kantrowitz team’s reports were accurate. Dr. Dennis assured the residents that he would support their continued training elsewhere and that he would be their advocate for credentialing issues.
NIH research funding …
Of the PPG 5 year funding of $3 million, 2 years of funding, over a million dollars, was unexpended. Because NIH policy stipulated that program-projects were not transferable, Kantrowitz discussed the Maimonides developments with Dr. James Shields, Chief of the NHI PPG staff. The NHI staff called a special meeting of the Heart Advisory Council. The Council’s members approved transfer of the 2-year remaining funds, all grant purchased equipment, the advanced instrumentation and equipment for the two-patient study unit, and specified that all hospital assigned IP be reassigned to Kantrowitz’s new institution. The approval was contingent on an NHI staff site visit to assure the adequacy of the successor facilities and administrative support for the Program Project. On May 29, 1970, MMC submitted its “Official Statement Relinquishing Interests and Rights to the Program Project Grant” to NIH.
The cooperative study …
Fully informed of developments, the Cooperative Study Investigators remained staunch. In 1969, The John A. Hartford Foundation, also fully informed, had approved funding of the Cooperative Study in the amount of $187 000 for 2 years. With Kantrowitz’s January 1970 resignation and June 30, 1970 planned departure, Maimonides agreed to administer the Hartford Foundation funds, enabling the Cooperative Study to begin at other centers in 1969.
Aftermath
Finding a new home …
By the late 1960s, the Executive Board of Sinai Hospital of Detroit (Sinai Hospital), a teaching affiliate of Wayne State University’s Medical School, had decided the Hospital would offer open heart surgery In preparation for the new service, Sinai Hospital had constructed a new surgical wing and a new research building. Dr. Alex Walt, Chair of Surgery at Wayne State University School of Medicine, asked Dr. Dennis to recommend candidates to lead Sinai’s program. Dr. Dennis recommended Kantrowitz.
Sinai Hospital appointed Kantrowitz as Chair of Cardiovascular Surgery, Dr. Joseph Krakauer as Associate Chair, and the eight surgical residents to its house staff. Dr. Walt appointed Kantrowitz Clinical Professor of Surgery at Wayne State. Subject to NHI approval, Sinai Hospital agreed to sponsor the program project, employ the research staff, and administer the PPG’s remaining funds. Dr. Rosalie Silverberg led an administrative site visit from NHI. The site visitors approved Sinai Hospital as an appropriate setting for the PPG. Remaining PPG funds, all NIH grant-purchased assets, and grant-supported intellectual property were reassigned to Sinai Hospital of Detroit. Within 1 month, an open-heart procedure was performed at Sinai Hospital. The IABP Cooperative Study continued with the inclusion of Sinai Hospital enrolled patients.
Fifteen months after the group arrived at Sinai Hospital, on August 10, 1971, a second-generation LVAD, the dynamic aortic patch, was implanted in a 63-year-old, terminally ill man 29. It was essentially a permanent IABP, this time cross-fertilized from the IABP (Fig. 16) 30. The dynamic aortic patch preclinical experiments had begun at Maimonides 31. The U-shaped blood pump was redesigned to be an elliptically shaped balloon implanted in the wall of the descending thoracic aorta, obviating the U-shaped device need for graft attachment to the aorta.
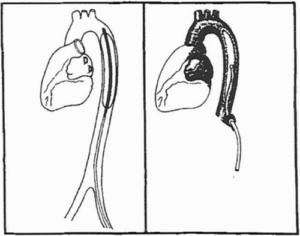
Figure 16
On September 18, 1971, 39 days after the implant surgery, the patient was discharged to home (Fig. 17). He went home with four drive units: (i) an 11″ bedside cube; (ii) a vest-worn unit; (iii) a wheelchair unit; and (iv) a suitcase unit. He enjoyed a good recovery for 3 months. At that time, an infection originating at the drive line entry site led to sepsis and death. He was the first chronic heart failure patient to be discharged to home with an LVAD designed for long-term use, i.e., “destination therapy.”

Figure 17
Haskell Shanks leaving Sinai Hospital of Detroit on September 18, 1971. He was the first person to be discharged to home with a left ventricle assist device intended to remain in the body and for the patient to be ambulatory.
Photograph courtesy of the Detroit News, Detroit, MI, USA.
After a second patient followed a similar course, Kantrowitz, with investigative dermatologists at the University of Michigan, began research in the late 1970s on a tissue-engineered percutaneous access device. The access device was precoated with autologous fibroblasts to inhibit epithelial downgrowth, reducing the risk of infection originating at the site of drive line skin penetration 32, 33. This became a major effort, driven by Kantrowitz’s conviction that, to the extent possible, electromechanical components to power and control the interruptible blood pump should be extracorporeal where, he said, “they can be replaced with a ‘loaner’.”
When long-term studies in Yucatan swine indicated that the tissue-engineered percutaneous access device remained stable and infection free, Kantrowitz sought permission to conduct a Feasibility Study of the CardioVad System in terminally ill heart failure patients. He obtained private sector funding and collaborators at three sites: Drs. Larry Stephenson and Joshua Wynne at Harper Hospital in Detroit, Dr. William Piccione at Rush Presbyterian in Chicago, and Dr. Valluvan Jeevanadam at the University of Chicago who reported the clinical experience with a “permanent intraaortic balloon pump” 34.
Allen Kantrowitz, a neurosurgeon and long-standing intimate colleague of his father, foresaw the joining of the percutaneous access device with the IABP. In 2008, he was issued a patent for a “Long-Term Ambulatory Intra-Aortic Balloon Pump with Percutaneous Access Device” 35.
The Cooperative Study report on 87 patients appeared as the lead article in the New England Journal of Medicine on May 10, 1973. Dr. Steven Scheidt, one of Dr. Killip’s Cardiology fellows at Cornell, was first author of the report, which concluded: “In patients who die with cardiogenic shock, more than 40 per cent of the left ventricle is destroyed often from a combination of old and new infarcts. Several workers have suggested that the areas of infarction gradually enlarge as the circulatory system deteriorates. A vicious circle may be engendered, with poor myocardial perfusion acting to enlarge areas of infarction, which then further compromises perfusion. Although adequate circulatory support is afforded by the balloon, improvement in survival is precluded by the presence of overwhelming and irreversible myocardial damage.”
“Our experience does not suggest that an appreciable decrease in the mortality from cardiogenic shock would result from more widespread use of the balloon or other circulatory support devices after the onset of shock. Rather, we recommend clinical evaluation of the hypothesis that the massive myocardial damage invariably associated with shock could be prevented or limited by intra-aortic balloon counterpulsation” 36.
On the technical side, in 1985, Aisin Seiki Co., Ltd. of Kariya City, Japan, invited Kantrowitz to design an IABP for the Japanese market. Aisin Seiki, a Toyota Family Company, supplied transmissions to the Toyota Motor Company and, at that time, to Volvo and Rolls Royce as well. From the first, Kantrowitz had envisioned the IABP as a closed-loop system with pressure sensing and electrocardiograph signal processing capability. In the late 1960s and early 1970s, the laboratory had a PDP15 computer. Dr. Mitchell Moskowitz and Arthur Ciarkowski (who later joined the FDA) struggled mightily to write the necessary programs, but the computer capability was insufficient. Commercial IABP drive units were being adjusted manually.
Kantrowitz told the Aisin Seiki Board of Directors that he was not interested in a “me too balloon pump.” He said he would be “interested in working on a closed-loop system for cardiac assistance” as he had originally imagined. “A closed loop, fully automatic system would provide more effective cardiac assistance and … the enabling computer capability had become available to do it.” The collaboration resulted in the first closed loop, fully automatic IABP—introduced to the Japanese market in April 1992 at the 23rd Annual Meeting of the Japanese Society of Cardiovascular Surgery in Sendai-city, Japan, by its distributor: Senko Medical (Fig. 18). Both Datascope and Arrow had shown strong interest in this first fully automatic IABP; however, each later developed an automatic system.

Dr. Adrian Kantrowitz presenting tracings made by the Aisin Seiki CORART, the first fully automatic intra-aortic balloon pump to Mr. Minoru Toyoda, CEO of the Company, March 5, 1986. Jean Kantrowitz and Mme. Ritsuko Toyoda were present.
A Plausible Answer to “Why the Exodus?” …
There was probably not a single cause for the mass move from Brooklyn to Detroit. The most likely root cause was Kantrowitz’s application for, and award of, the PPG, which provided for the two-bed clinical study unit for circulatory assist patients. The handsomely designed unit had been officially opened in January 1969. Within 6 months, Kantrowitz was informed of the Ad Hoc Subcommittee review of IABP patients.
At that time, NIH policy was that a PPG was not transferable: If the Principal Investigator of the PPG left the host institution, the institution was committed to replace the Principal Investigator and to continue the program-project. The clinical study unit, all PPG purchased equipment and unexpended funds would remain at Maimonides Hospital. As Kantrowitz had a thriving program, the prevailing thinking among the staff may have been that he was not likely to leave Maimonides.
Did this NIH policy regarding PPGs suggest a strategy to acquire an NIH-funded general CRC for Maimonides? Did Kantrowitz’s colleagues seek means so intolerable to him that he would leave MMC? In that case, Kantrowitz would have to abandon the PPG funds, the two-bed clinical study unit, and all equipment purchased with PPG funds at Maimonides.
In such a hypothetical strategy, only a new Principal Investigator would have to be named by the Hospital. When Kantrowitz’s colleagues voted to suspend the IABP study at MMC, as expected, Kantrowitz’s resignation quickly followed. However, contrary to expectation, the NIH Staff sought an exception to their Program-Project “nontransferable policy” and did everything possible to successfully facilitate continuation of the PPG in a new setting.
In Retrospect
The process of bringing an innovative device from bench to bedside takes place now in a very changed environment. At the NIH Agency level, during the 1960s, transfer of a PPG was precluded, a constraint since relaxed. The game changers were two federal initiatives: In 1976, FDA regulation of medical devices in the USA went into effect. In 2004, the NIH Roadmap was launched.
The FDA … brought with it a sequence of approvals for a medical device born of an understanding of the iterations needed to bring a device from concept to bedside. No matter how extensive the bench and preclinical animal studies, they cannot reveal all the challenges presented by patients. Device developers could now apply for an investigational device exemption to conduct a Feasibility Study. Readiness of the device for test in human subjects would be judged by the FDA staff and its expert advisors. A Feasibility Study allows evaluation of safety in a small number of patients. It allows developers to go back to the drawing board, modify, and retest a device until its readiness warrants use in larger numbers of patients in a randomized trial. The Maimonides initial trial was a Feasibility Study. The Cooperative Study was a multicenter Feasibility Trial. Both the NHI staff and the Medical Research Committee at MMC had insisted upon a randomized trial after relatively few patients in the hands of a single team at one medical center. At that juncture, a randomized trial could well have miscarried the innovation.
The question of whether innovation can thrive in a community hospital is now largely moot. The NIH Roadmap provides for “bold initiatives … to pursue compelling opportunities.” It encourages institutional collaboration. It is no longer essential for an innovator to be dependent on the oxygen of one institution. Translational research reduces the risk of road blocks such as those encountered during the introduction of the IABP. The Roadmap is creating a culture in which initiatives, emanating from a variety of settings, including a community hospital, can thrive 37. The turbulent path of the IABP from bench to bedside was an episode of translational medicine that occurred more than four decades before translational medicine became formalized. Innovative devices, including next generations of LVADs placed using minimally invasive surgical (MIS) techniques, counterpulsating ambulatory balloon pumps, and off-the-shelf tissue engineered percutaneous access devices, including those of yet another A. Kantrowitz (Allen), are the promising beneficiaries of this now recognized discipline, Translational Medicine.FDA regulation …
NIH roadmap …
A Postscript: Heroes of the IABP Story
The perilous journey of the IABP from bench to bedside was supported by many heroes largely unacknowledged in later accounts. They include:
- Dr. Clarence Dennis—Dr. Dennis was not a physically large man but, for strength of character, none stood taller. Following the Research Committee’s suspension of the IABP clinical trial at MMC, Dr. Dennis personally adjudicated the integrity of the IABP study patient reports. He intervened with MMC to protect the medical residents committed to the Assisted Circulation Research Program, and he actively sought and found a home for the program.
- The NIH Staff—The impassioned salvage of Kantrowitz’s assisted circulation PPG did not take place in a vacuum. The NIH staff’s dedicated, skillful efforts took place against the backdrop of national focus on the need for improved treatment of heart failure. Inspired by Dr. Michael DeBakey and muscled into policy by President Lyndon Johnson, NIH was pursuing proactive means of stimulating mechanical methods of replacing and/or assisting the failing heart. The program-project, “Studies in Assisted Circulation” was in step with the national direction. It was among the first, if not the first, investigator-initiated, comprehensive, multidisciplinary research and development program in mechanical circulatory assistance, encompassing an array of projects to take concepts from bench to bedside (Fig. 19). The NIH program-project staff—Drs. Robert Ringler, James Shields, Rosalie Silverberg, Malvina Schweizer, and Mr. James Pike—moved mountains to keep the assisted circulation PPG alive. Such a huge effort could not have happened without the encouragement of Dr. Theodore Cooper, then Director of the NHI.
- The Cooperative Study Investigators—These outstanding physicians were a seasoned group accustomed to and unfazed by institutional politics. Dr. Thomas Killip III rode herd on the project. Dr. Stephen Scheidt of Dr. Killip’s Cornell group prepared the cooperative study report that appeared as the lead article in the May 10, 1973, issue of the New England Journal of Medicine. The Data Coordinating Center at St. Vincent’s Hospital, New York, which included Drs. Peter Fleming, Hiltrud Mueller, Stephen Ayres, E. Foster Conklin, and Stanley Gianelli, did yeoman service.
- The Maimonides Assisted Circulation Staff—The heavy lifting in the experimental surgery laboratory and study patient care was the work of talented, dedicated people: Dr. Joseph Krakauer never left the bedside of a study patient; Paul Freed came to do his master’s thesis and spent his entire career in the Assisted Circulation Program. Paul always said that something could not be done and then proceeded to do it. Dr. Tetsuzo Akutsu and Dr. Dov Jaron managed the experimental surgery laboratory with a tight rein, each making their own important contributions, alongside Research Associates from all over the globe.
- Several members of the team were not able to make the move to Detroit for personal reasons: Editorial Assistant Michael Meyer; Research Nurse Sheila Berg; and Prototype Maker Sidney Wolvek. Sidney’s prior employment had been with Abercrombie and Fitch, where he built model ships for wives who commissioned models of their husbands’ yachts as birthday gifts. Sidney was taught to make balloon pumps by Paul Freed. He later went to work at Datascope. There Sidney designed the furled balloon pump enabling percutaneous insertion of the IABP by cardiologists, rather than surgeons, an innovation that expanded the use of IABP overnight.
- The Residents—Incredibly, at the highest career risk, eight surgical residents—George Hines, MD, Michael Ciborski, MD, Peter Goodman, MD, Claudio Titone, MD, Alejandro Aris, MD, Marc Lipsius, MD, George Zorzi, MD, and Steven Phillips, MD—refused to stay on at Maimonides. Determined to remain with Kantrowitz, these Residents had no way of knowing whether that was even a possibility. When the Sinai Hospital home was found for the entire group, they picked up their families and moved to a new medical center and new life in the Midwest.

Figure 19
The Program-Project allowed rapid movement between counterpulsating concepts. Dr. Kantrowitz is holding a balloon pump. A variety of VAD prototypes are on the desk.
Acknowledgment
The author is indebted to many colleagues and friends who confirmed or refreshed my recollections, or by their silence, thundered “cut it down”, among them: Paul Freed , Drs. Dov Jaron, Robert Bartlett, George Pantalos, Steven Phillips, Keith Aaronson, and John Watson. Thanks are due to Elaine Taro for research and over-the-top general assistance, to Dr. Lisa Rosenbaum for encouragement that the “story was worth telling” and to Dr. Susan Speaker, Historian at the National Library of Medicine, Curator of the Adrian Kantrowitz Collection at the National Library of Medicine for their invaluable assistance.
Footnote
- *Maimonides Hospital was renamed Maimonides Medical Center in 1967.


