John-Peder Escobar Kvitting1,2 | Bansi Koul3 | Odd R. Geiran1,2
1Department of Cardiothoracic Surgery, Oslo University Hospital, Rikshospitalet, Oslo, Norway 2Institute of Clinical Medicine, University of Oslo, Oslo, Norway 3Department of Cardiothoracic Surgery, Skåne University Hospital, Lund University, Lund, Sweden
Correspondence
John-Peder Escobar Kvitting, MD, PhD, Department of Cardiothoracic Surgery, Oslo University Hospital, Rikshospitalet, Post Office Box 4950, Nydalen 0424, Oslo, Norway.
Email: jpkvitting@gmail.com
Abstract
In 1985, the surgical team led by Bjarne Semb implanted the first total artificial heart (TAH) in Europe, and the following year the first successful bridge to transplant in Europe using the Symbion J-7/ 100 TAH. Together with the clinical experiences of colleagues in the United States, these early cases preceded the subsequent development of scores of mechanical assist devices to treat advanced heart failure. Semb proved to have the pioneering spirit needed to use the early generation of a TAH, but these early implants also generated much controversy in the medical community as well as the general public.
Bjarne Kristian Holtermann Semb was born August 3, 1939, in Namsos, Norway. Semb completed his medical studies at the Ludwig Maximilian University of Munich, Germany, in 1963, and he completed his training in general and thoracic surgery at Rikshospitalet in Oslo.
Semb defended his doctoral thesis at the University of Oslo in 1971 with the title: Cardiac Transplantation: An Experimental Study in Dogs (Figure 1A).1 Christiaan Barnard was a co-author on two of the articles in Semb’s doctoral thesis (Figure 1B). Before Barnard performed the first human heart transplantation in South Africa, Barnard and Semb had formed part of the kidney transplant team in 1966 under the leadership of David M.Hume at the Medical College of Virginia in Richmond, VA. Together with Hume, Semb published his first academic article in 1968 on allogenic lymphocytes in an experimental kidney model.2 Before completion of his training in Oslo as a cardiac surgeon he did extensive experimental research on regulators of gastric mucosal flow. After his return to Rikshospitalet, he did clinical studies on the early version of the Hall-Kaster disc valve prosthesis.
In 1984, Viking Olov Björk retired from his dual position in Stockholm, and Semb was recruited for his knowledge of heart transplantations and was appointed head of the Department of Cardiothoracic Surgery at the Karolinska Hospital as well as professor of Thoracic Surgery at the Karolinska Institute.
1 | THE FIRST IMPLANTATION OF A TOTAL ARTIFICIAL HEART IN EUROPE
At the time of Semb’s appointment in Stockholm, the concept of brain death was not widely accepted among the general public and heart transplantations were considered controversial in Sweden. Prior to 1984, the authorities had banned Björk from launching a heart transplantation program. Swedish patients in need of a heart transplant were cared for at the Harefield Hospital in England, in collaboration with Sir Magdi Yacoub.
The Swedish National Board of Health and Welfare gave Semb permission to perform three heart transplants


FIGURE 1 The cover page of Bjarne Semb’s doctoral thesis from 1971 [A]. The two articles in Semb’s doctoral thesis which were co-authored with Christiaan Barnard [B] [Color figure can be viewed at wileyonlinelibrary.com]
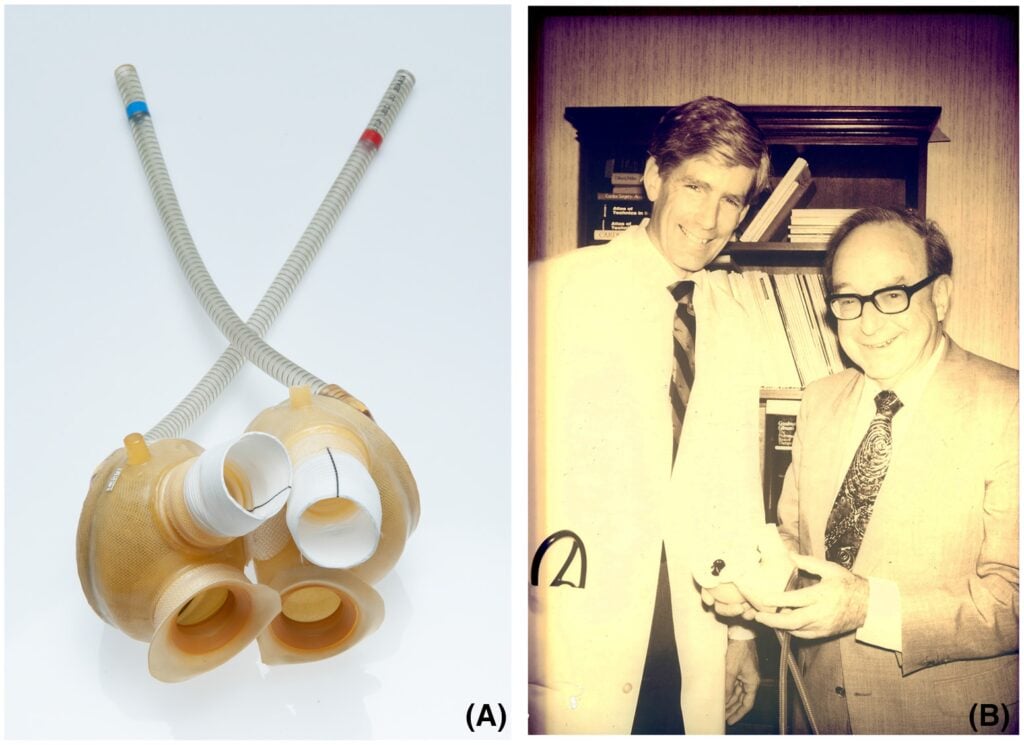
FIGURE 2 The Symbion J-7/ 100 total artificial heart (TAH) [A]. Karl Viktor Hall holds the Symbion J-7/100 TAH together with William DeVries [B] [Color figure can be viewed at wileyonlinelibrary.com]
with donor hearts procured outside of Sweden. To prepare for these transplants, Semb sent staff members from the Karolinska Hospital to train at Harefield Hospital. Due to the political and legal constraints, Semb worked to establish alternative treatments for Swedish patients with advanced heart failure. He contacted Willem Kolff, who had previously developed the first renal dialysis machine for clinical use, in Salt Lake City, Utah. Kolff had, in collaboration with Robert Jarvik, developed a total artificial heart (TAH) for clinical use and commercialized it under the name Symbion J-7/100 TAH (originally called the Jarvik-7) (Figure 2A).
The Medtronic-Hall tilting disc heart valve prosthesis was used in the later version of the Symbion J-7/100 TAH, and Semb had in his career in Oslo published on the clinical outcome of the Medtronic-Hall valve. The Medtronic-Hall valve had been chosen for the Symbion J-7/100 TAH due to its excellent durability and low thrombogenicity.3 The Symbion J-7/ 100 TAH had been approved by the FDA in 1981 for human use as an alternative to heart transplantation as an IDE (investigational device exemption), and on December 2, 1982 the Symbion J-7/100 TAH was implanted by the surgical team led by William DeVries at the University of Utah (Figure 2B). The first patient died 112 days post-operatively due to a stroke and a device infection. In the United States, four additional patients received the Symbion J-7/100 TAH, but with disappointingly short survival.4
In early 1985, a team from Stockholm received extensive training with the implantation of the Symbion J-7/100 TAH on calves in Utah. This training sojourn was followed by several reciprocal visits between the two centers to improve the implantation procedure in the human chest.5 Furthermore, a mock circulation device was used to refine the tuning of right and left ventricular function to improve the transition from cardiopulmonary bypass to the Symbion J-7/100 TAH.6
In the spring of 1985, a patient suitable for a TAH was presented to the surgical team at the Karolinska Hospital. The patient was a 53-year-old man with ischemic cardiomyopathy in NYHA class IV, with co-morbidities in the form of liver cirrhosis, reduced kidney function as well as pulmonary hypertension. The patient had been evaluated at Harefield Hospital for heart transplantation but was found unsuitable for this therapy. After further evaluation in Stockholm, however, he was accepted to receive a TAH as destination treatment.
The implantation of the first TAH in Europe took place on April 7, 1985, and Semb was assisted by Bansi Koul and Jan Liska.7 Initially, the TAH implantation went smoothly, but when the patient was weaned from cardiopulmonary bypass to the TAH, acute fulminant pulmonary edema occurred after only a few beats with the TAH. The surgical team concluded immediately that the left atrium pressure catheter had dislodged and was impinging the movement of the tilting disc valve in the mitral position. The TAH was stopped and the left ventricle was disconnected from the left atrium and the catheter was removed to ensure the free movement of the tilting disc valve. The next weaning attempt proceeded uneventfully, but there was a problem with continuous bleeding from the back of the left atrium, presumably from a stretch in the atrial suture line after the removal of the pressure catheter. The operation lasted 14 h, mostly due to the bleeding from the left atrium.
The patient was extubated on the second post-operative day and was in good mood (Figure 3A,B). Despite a continuous heparin administration in combination with warfarin and acetylsalicylic acid in a low dose, the patient suffered a minor stroke on the post-operative day 132. Prior to this event, the patient was fully mobilized with daily walks inside the clinic and outside the hospital to restaurants and cinemas, using the completely integrated wearable TAH-drive unit (weight of 5 kg) designed by dr. Peter Heimes (Figure 3C). This was the first time in the world that the portable Heimes driver was used clinically. The unit consisted of an electric motor-driven ball screw/piston. A microcomputer allowed adjustments of drive parameters based upon an indirect preload calculation: systolic duration could be varied between 40% and 60% of cardiac output of 2 to 11 L/min with a stroke rate between 50 and 130 bpm. A rechargeable battery lasting up to 4 h powered the wearable unit.8
The patient was planned to be transferred to his own apartment when he suffered from the first stroke. After the first stroke, the patient was rehabilitated enough to walk with support, but he could not leave the hospital as initially planned.
About 200 days post-operatively, the drivelines through the abdomen became infected, and despite extensive treatment with antibiotics, the patient died 220 days after the operation due to sepsis and multiorgan failure. Despite this setback, the experience had demonstrated the potential for recovery of organ function with the use of a TAH. As stated by Semb: “after we put in the Jarvik-7, his renal function returned to normal and, to a large extent, his liver function as well. This let us feel that he would now be able to withstand a transplant operation”.9
The TAH implantation as destination therapy, the third in the world and the first in Europe, garnered massive media attention from all over Europe, as well as the USA—not only as a medical breakthrough but for the particular nature of the patient, referred to as “Mr. X” in the media. The patient was referred to as “the gangster king,” and a headline in the New York Times on April 11, 1985, stated, “Sweden’s heart implant recipient linked to police and tax inquires”.10 Sweden prompted the introduction of a brain death law on January 1, 1988.
2 | THE FIRST BRIDGE TO TRANSPLANTATION IN EUROPE USING A TAH
In May 1986, the first successful bridge to transplantation in Europe using a TAH (the second in the world after
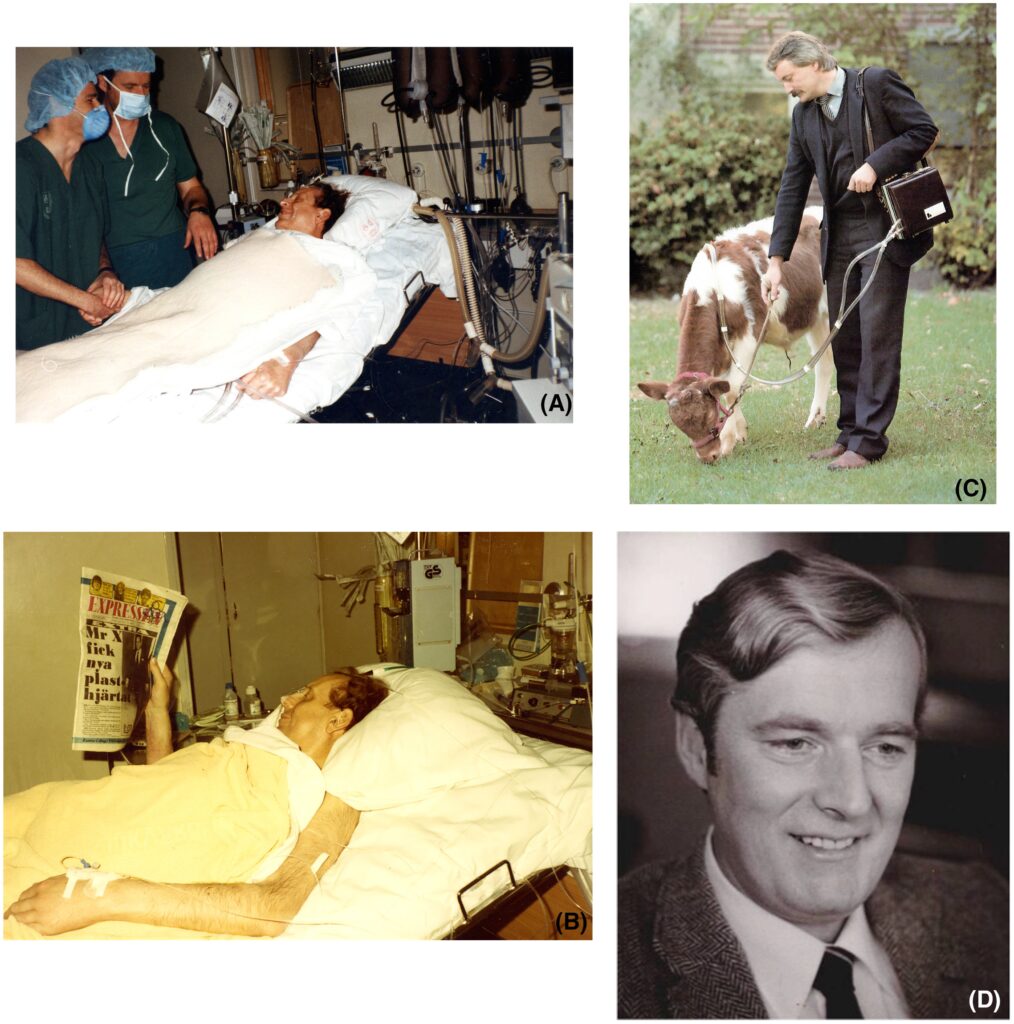
FIGURE 3 The patient after his extubation greeted by doctors Semb and Jarvik [A]. The patient in the intensive care unit reading about his own total artificial heart (TAH) operation in a local newspaper including the disclosures about his previous economic irregularities toward the Swedish state [B]. Dr. Peter Heimes and the completely integrated wearable TAH-drive unit for the Symbion J-7/100 [C]. Professor Bjarne Semb (1939–2017) [D] [Color figure can be viewed at wileyonlinelibrary.com]
the first was performed by Jack Copeland in Arizona), was performed by Semb and his team at the Karolinska Hospital. The patient was a 56-year-old man with idiopathic cardiomyopathy in pre-shock, and the Symbion J-7/100 TAH was used as a lifesaving procedure. Ten days post-operatively, the patient underwent successful heart transplantation. The patient died three years post-transplant due to a lympho-proliferative disease secondary to the immunosuppressive treatment. The third patient to receive the Symbion J-7/100 in Stockholm was a female patient from Germany with Uhl’s disease referred after failed surgical repair for a tricuspid insufficiency. The patient received a heart transplant after 9 days with the TAH, but she developed an acute rejection and then received a new TAH. After 180 days with the second TAH, the patient was transplanted again, but died intra-operatively due to failure of the new transplant, probably due to chest restriction related to extensive scar tissue in the chest cavity secondary to the TAH.
During the time period from 1985 to 1987, a total of nine heart transplants, with donors from outside of Sweden coordinated through Eurotransplant, were performed in Stockholm. Two of these cases were transplanted from the TAH as a bridge to transplant.
The implantation of TAHs and heart transplantations generated several ethical issues that led to an open conflict both in the department and in the news media. The conflict ended with Bjarne Semb leaving his position at Karolinska Hospital, but continuing as a professor at the Karolinska Institute. After leaving Stockholm, Semb worked for several years in Wisconsin at the Milwaukee Heart Associates and Medical College (Figure 3D).
In parallel with the development of TAHs in the USA, important and substantial contributions to the field were made behind the Iron Curtain. In the former Soviet Union, collaboration between professor Valery Ivanovich Shumakov and dr. Michael DeBakey resulted in the TAH called “Poisk-10M.” From 1987 to 1991, 17 end-stage heart failure patients (13 in the Soviet Union and 4 in Poland) received the “Poisk-10M” TAH.11 Furthermore, at Brno Research Center in former Czechoslovakia a comprehensive TAH program was established. The TNS Brno VII TAH was implanted in animals with record-breaking survival times in a calf and a goat of 314 and 188 days, respectively. Important knowledge of the long-term effects of a TAH was elucidated by these animal experiments. The first clinical implantation of the TNS Brno VII TAH was done in 1988 and a total of 7 clinical applications were done with this device.12
3 | ETHICAL CONTROVERSIES OF THE FIRST TOTAL ARTIFICIAL HEARTS
The rapid development of technical and surgical aspects of the TAHs generated several different medical and political disputes in the treatment of patients with advanced heart failure. The first implantations of a TAH received massive attention in the news media, and the subsequent discussions became very heated. The many clinical problems associated with the early devices strained the reputation of TAHs and promoted some to advocate stopping all use of the devices until improvements were made to the designs.13
An international meeting arranged by the Foundation for American Communications on TAHs in Washington, DC in 1985 was attended by Semb together with DeVries and Copeland. Semb commented on his own experience with the Symbion J-7/100 in Sweden. In his opinion, the status of TAHs at that time was not sufficient to recommend a TAH as an alternative to heart transplantation, but that TAHs could be used as a bridge to transplant. Shortly thereafter, the FDA banned the use of the Symbion J-7/100 as an alternative to heart transplantation. The surgical community was, however, not all in agreement with these conclusions, and DeVries championed the use of the contemporary TAHs as destination therapy. The early reports on the TAH had in some news media been given a negative image, and questions had been raised about the viability of the devices. Many of today’s controversies regarding the indications and use of left ventricular assist devices (LVADs) mirror the issues discussed at this meeting among the pioneers in the field of mechanical assist therapy for the failing human heart.
The early development and clinical experience with TAHs paved the way for the later development of LVADs and their widespread clinical use as a bridge to transplant, bridge to recovery, and destination therapy. Infections and thromboembolic complications that hampered the use of the first generation of TAHs remain the Achilles heel in newer generations of LVADs. In patients with advanced biventricular dysfunction, a TAH is still a durable alternative as a bridge to transplantation.14
Dr. Jack Copeland has stated the following about dr. Semb and the early use of the TAH (personal communication, February 28, 2022): “Dr. Semb was a pioneer in the use of the total artificial heart. He followed Dr. DeVries by implanting the Jarvik-7 TAH as a long term therapy for heart failure. The device worked well and the patient survived over 200 days. He went on to successfully use the TAH as a bridge to transplantation. He, like DeVries and myself (after our initial bridge to transplantation in 1985), were caught up in publicity that often obscured the landmark observation that the TAH was very successful at totally supporting patients suffering from irreversible, progressive, and life threatening biventricular heart failure.
Now over 2000 TAH implants have been done, mostly for bridge to transplantation with essentially the same device Semb used. Survivals to transplantation have been in the range of 70–80%, complications have been much reduced, and survival post-transplant has been 80% or better. And, the TAH has survived to start becoming appreciated as an important lifesaving technology for supporting and resuscitating patients that have “slipped through the cracks” for many years.15 Also, he may not have known that over 200 TAH recipients have lived over 1 year on TAH support and several have lived for over 6 years.
Dr. Semb should be remembered as a man with the vision to see that this was possible and the courage to do pioneering work. A pioneer is willing to risk his own reputation for a worthy cause. In my opinion, he was a great surgeon and a pioneer.”
Semb was very result-oriented and felt that the development of surgical techniques and the introduction of novel devices should preferably take place within a small specialized group and then be incorporated as a routine for the entire clinic. With this approach, however, he did not succeed in making his surgical staff feel genuinely involved with the new thinking and the novel procedures that to some extent became unique in the world. This line of conflict and the controversies that were generated with the first TAH implants have tainted the legacy of Semb. His groundbreaking contribution to the field of mechanical assists devices to treat terminally ill heart failure patients, however, should remain a milestone in the field of cardiac surgery. Bjarne Semb died on November 7, 2017.
ACKNOWLEDGMENTS
The authors acknowledge Michael T. Durheim, M.D., for his valuable help with the manuscript.
AUTHOR CONTRIBUTIONS
John-Peder Escobar Kvitting, Bansi Koul and Odd R.Geiran: Concept/design, drafting article, critical revision of the article, and approval of article.
ORCID
John-Peder Escobar Kvitting https://orcid.org/0000-0001-5648-7688
Bansi Koul https://orcid.org/0000-0002-3481-6556
Odd R. Geiran https://orcid.org/0000-0002-8075-7059
REFERENCES
1. Semb BK. Cardiac transplantation. An experimental study in dogs. Oslo: Scandinavian University Press (Universitetsforlaget); 1971.
2. Semb BK, Williams M, Hume DH. The effect of allogenic lymphocytes on the isolated perfused kidney. Transplantation. 1968;6(9):977–85.
3. Kvitting JP, Geiran OR. Karl Viktor Hall: from in situ vein bypass to the tilting disc heart valve prosthesis. Ann ThoracSurg. 2016;102(5):1756–61.
4. DeVries WC, Anderson JL, Joyce LD, Anderson FL, Hammond EH, Jarvik RK, et al. Clinical use of the total artificial heart. N Engl J Med. 1984;310(5):273–8.
5. Jarvik RK, DeVries WC, Semb BK, Koul B, Copeland JG, Levinson MM, et al. Surgical positioning of the Jarvik-7 artificial heart. J Heart Transplant. 1986;5(3):184–95.
6. Liska J, Semb BK. In vitro flow characteristics of the Jarvik-7 prosthesis with respect to filling pressure, stroke frequency, and systolic function. Eur J Cardiothorac Surg. 1989;3(6):515–20.
7. Koul BL, Liska J, Svensson J, Semb BK. Clinical application of the total artificial heart. Life Support Syst. 1985;3:195–9.
8. Heimes HP, Klasen F. Completely integrated wearable TAH-drive unit. Int J Artif Organs. 1982;5(3):157–9.
9. Marwick C. Pondering past, future of implantable heart. JAMA. 1985;254(23):3291–2.
10. The New York Times. April 11, 1985, Section A, p. 18.
11. Gautier SV, Shevchenko AO, Itkin GP, Zakharevich VM,Poptsov VN, Drobyshev AA, et al. Artificial heart in Russia:past, present, and future. Artif Organs. 2021;45(2):111–4.
12. Dostál M. From womb to tomb: the Brno TAH story. Artif Organs. 1995;19(6):551–6.
13. Kolata G. Surgeons disagree on artificial heart. Science. 1985;230(4727):786–7.
14. Chung JS, Emerson D, Megna D, Arabia FA. Total artificial heart: surgical technique in the patient with normal cardiac anatomy. Ann Cardiothorac Surg. 2020;9(2):81–8.
15. Copeland J, Langford S, Giampietro J, Arancio J, Arabia F. Total artificial heart update. Surg Technol Int. 2021;39:243–8.

